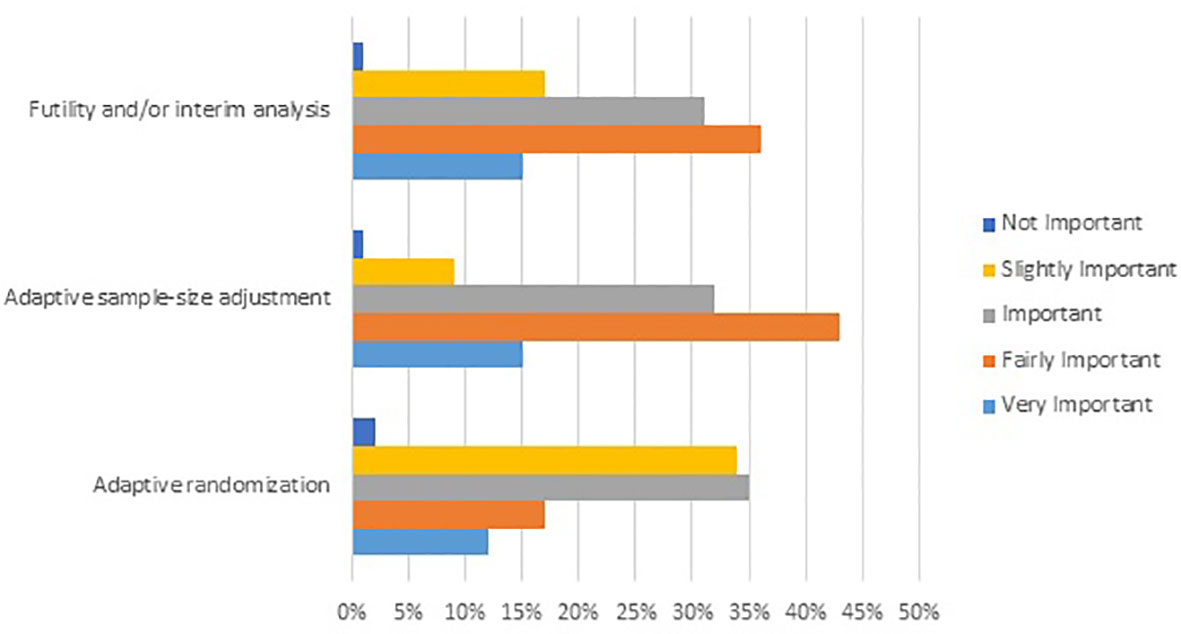
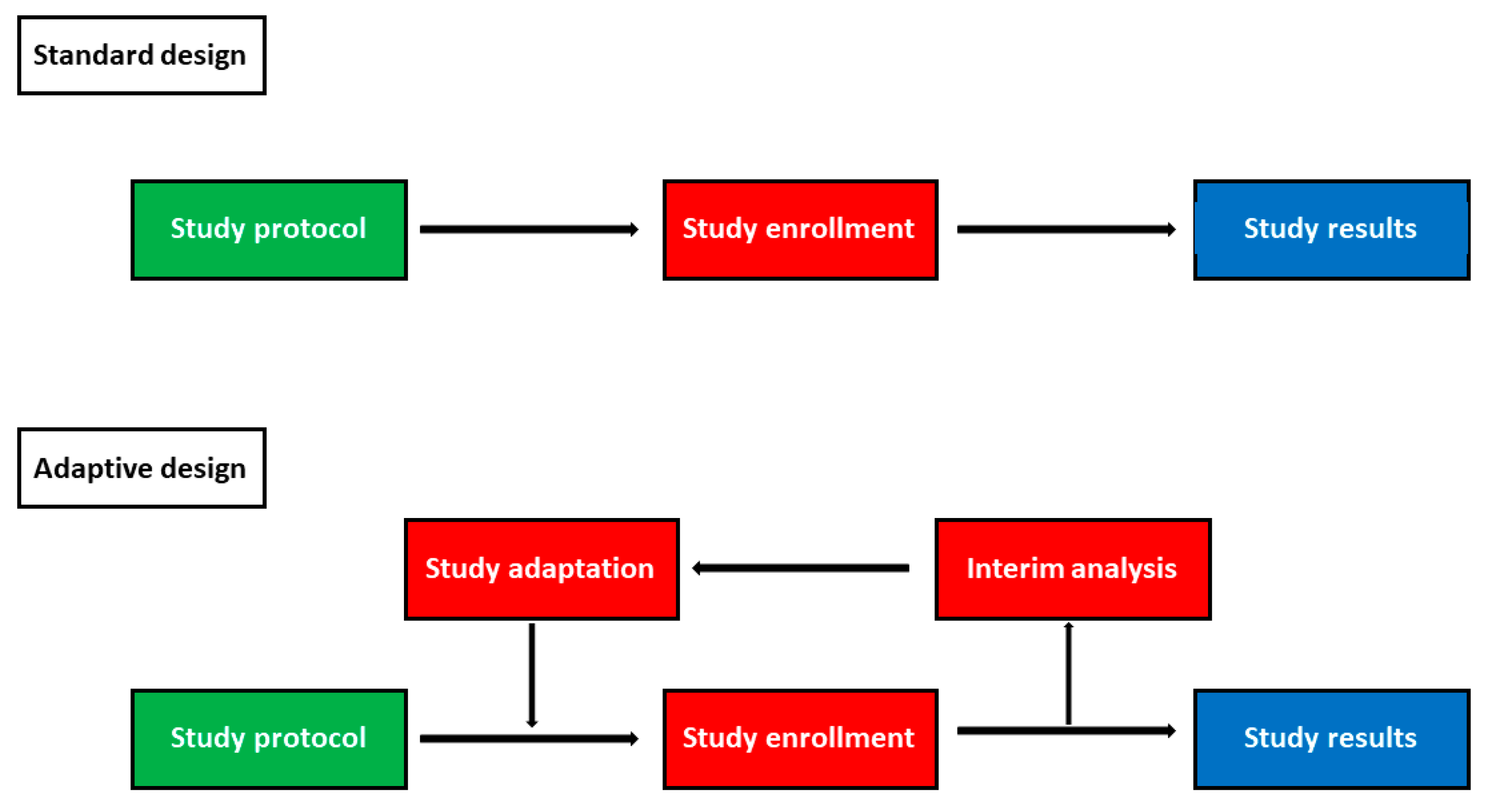
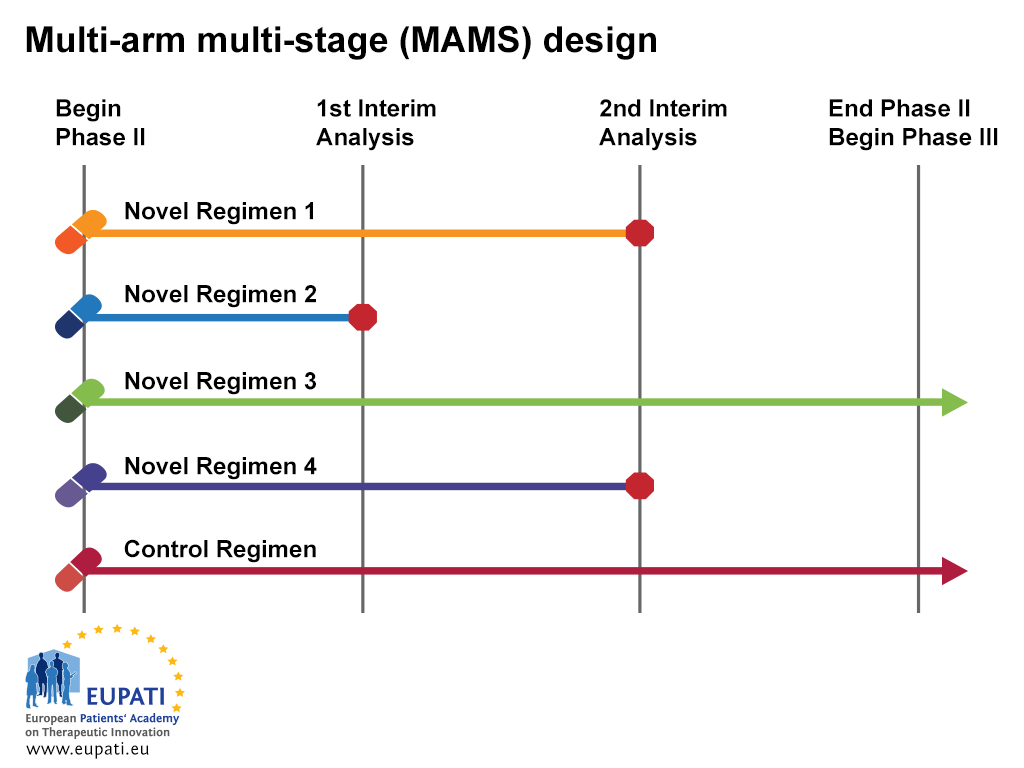
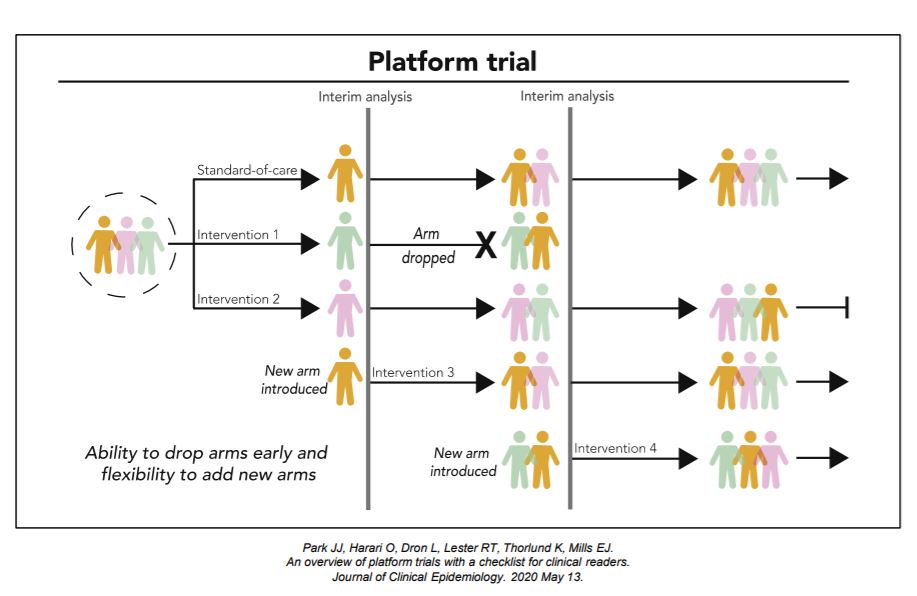
Development and Evaluation of a Simulation-Based Algorithm to Optimize the Planning of Interim Analyses for Clinical Trials in ALS | Neurology

Clinical Trials - analysis End-points, planned and unplanned secondary analyses, interim analyses, stopping rules Dick Menzies, MD Respiratory Epidemiology. - ppt download
Evaluating Clinical Trial Designs for Investigational Treatments of Ebola Virus Disease | PLOS Medicine

The use of interim data and Data Monitoring Committee recommendations in randomized controlled trial reports: frequency, implications and potential sources of bias | BMC Medical Research Methodology | Full Text
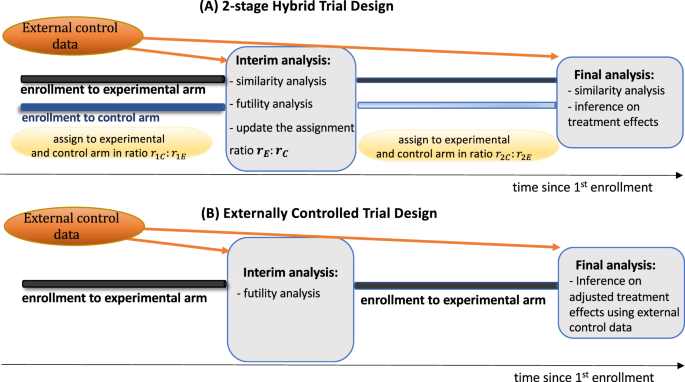
The design and evaluation of hybrid controlled trials that leverage external data and randomization | Nature Communications

PPT – Interim Analysis of Clinical Trial Liying XU CCTER, CUHK PowerPoint presentation | free to download - id: 3c7e9a-ZjcxY